What You Missed at the 7th Obesity & NASH Drug Development Summit?
The 7th Obesity & NASH Drug Development Summit was a one-stop shop to capture the industry’s most exciting developments in tackling metabolic syndromes
The fluctuating obesity and NASH landscape and failures has herded many former players away, but leading biopharma are determined to thrive in this ever-growing market.
With the obesity market expected to grow to $50 billion by 2030, the 7th Obesity & NASH Drug Development Summit was a timely meeting, uniquely positioned to capture all the latest breakthroughs and research you need to join the race and commercialize your GLP-1 RAs, tri-agonists, dual agonists, tetra-specific drugs, glucocorticoid receptor modulators and more!
The evolution in digital pathology, artificial intelligence, omics, multifunctional molecules, non-invasive biomarkers, target discovery and liver models deemed 2023 an exciting year to bring the metabolic community closer to even more approvals.
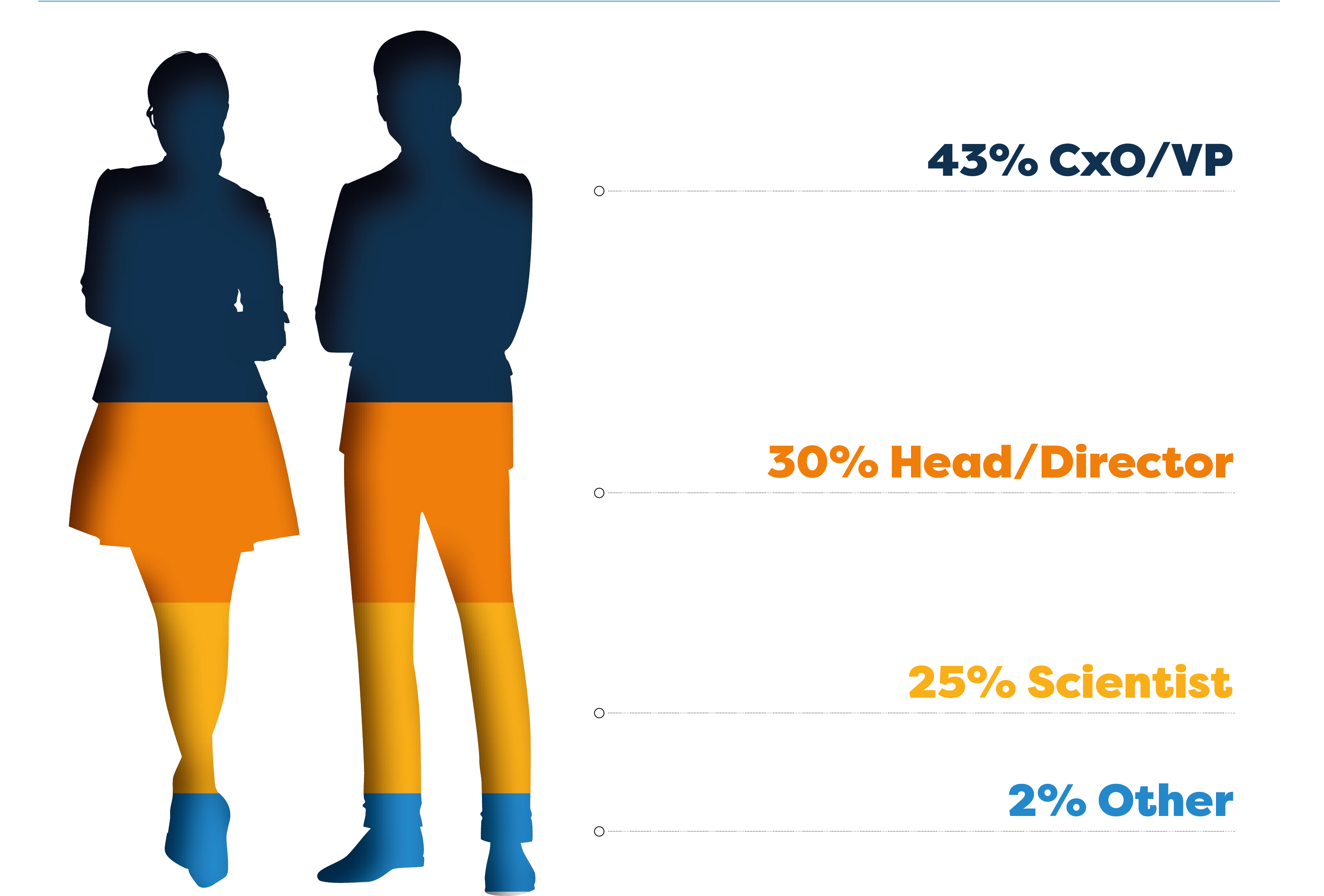
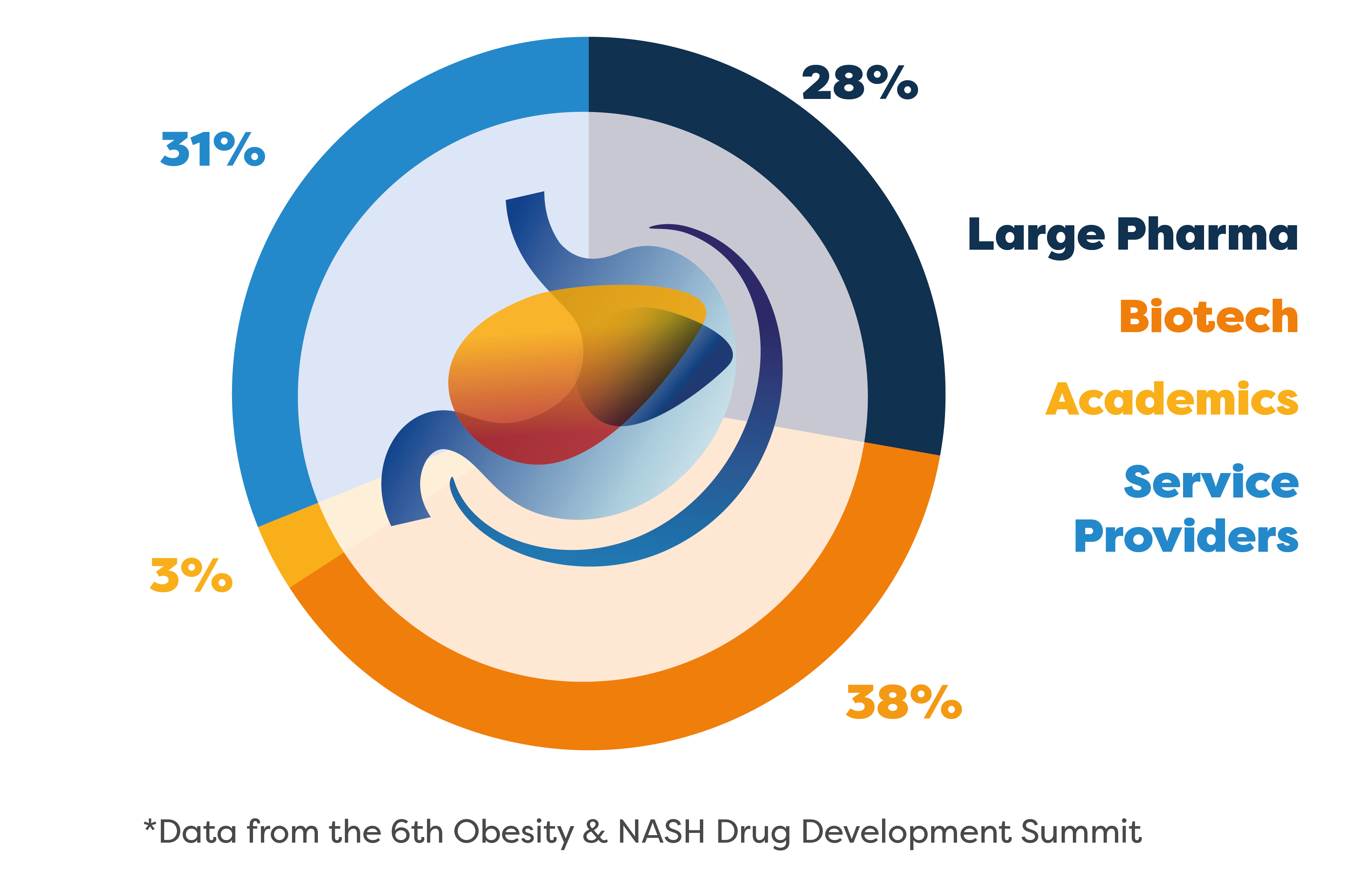
The 7th Obesity & NASH Drug Development Summit was an opportunity to hear from 12 new speaker companies and 34 new speakers over 3 days jam-packed with all the latest end-to-end focal updates!
2023 Highlights:
Who Did You Miss?
What Your Peers Had to Say:

"The quality of the presenters was excellent"

“Excellent speakers, good topics on agenda and networking opportunity to meet likeminded group”

“Ample opportunities to make personal connections and discuss findings and new perspectives in person with fellow conference goers”

"Attending this meeting provides an important opportunity to network with leading researchers and colleagues in Industry and keep up with cutting edge research and developments in NASH"




